# Hot Search #
Along with life’s evolution, the immune system has become increasingly sophisticated compared with 450 million years ago, when fish took the lead in deriving an adaptive immune system based on T- and B-cells. This system has been inherited and developed during the long evolutionary process, thus becoming the criterion for the immune defense of quadrupeds. However, evidence from comparative genomics illustrate that the critical T-cells and B-cells of adaptive immunity appear to have undergone distinct evolutionary patterns.
To illustrate further, take a look at how the B-cell immunity has gradually improved with the species evolution, which is not only reflected in the sequential appearance of new immunoglobulins and germinal centers, but also in the successive acquisition of antibody affinity maturation, type switching and other mechanisms.
By contrast, during the evolution from teleosts to tetrapods, the types and core components of T-cells keep the same form. In retrospect of the 450-million-year evolutionary history, the pressure of natural selection has caused five mass extinctions, which caused enormous changes in immune organs, living environments, and the dietary structure of quadrupeds and fish. In response to these challenges, it is not difficult to understand the evolution of B-cell immunity, while the stable inheritance of T-cell immunity seems more mysterious to researchers.

An ECNU research team led by Prof. Yang Jialong from the School of Life Sciences published a research paper: “Glutamine metabolism underlies the functional similarity of T cells between Nile tilapia and tetrapod”. The paper was published in Advanced Science, revealing the evolutionary rules of T-cell immunity. The authors take tilapia, Xenopus laevis, chicken, mouse and other animals as research objects, trying to answer two scientific questions: (1) Whether the stable evolution hypothesis of T-cells at the comparative genome level is reflected in the immunological process, function and regulation mechanism? (2) What are the underlying driving forces that maintain the stable evolution of T-cell immunity?
The authors first used the lower vertebrate tilapia as a model to describe the profile of early T-cell immunity. Tilapia’s T-cells, through dynamic proliferation and apoptosis, have formed a typical primary and memory immune response mechanism, of which the complete activation of the fish T-cells depends on the synergistic effect of the first and second signals, and is co-regulated by Ca2+-NFAT, MAPK/ERK, The NF-kB and mTORC1 pathways. Next, the selective depletion model suggests that IgM+ B cells can present antigens to T-cells, ensuring the activation and proliferation of T-cells. Functionally, the fish T-cells feature the cytotoxic function of specifically killing targeted cells, and can assist with the proliferation and antibody secretion of IgM+ B-cells. These results suggest that core strategies of T-cell immunity are well established in teleosts.
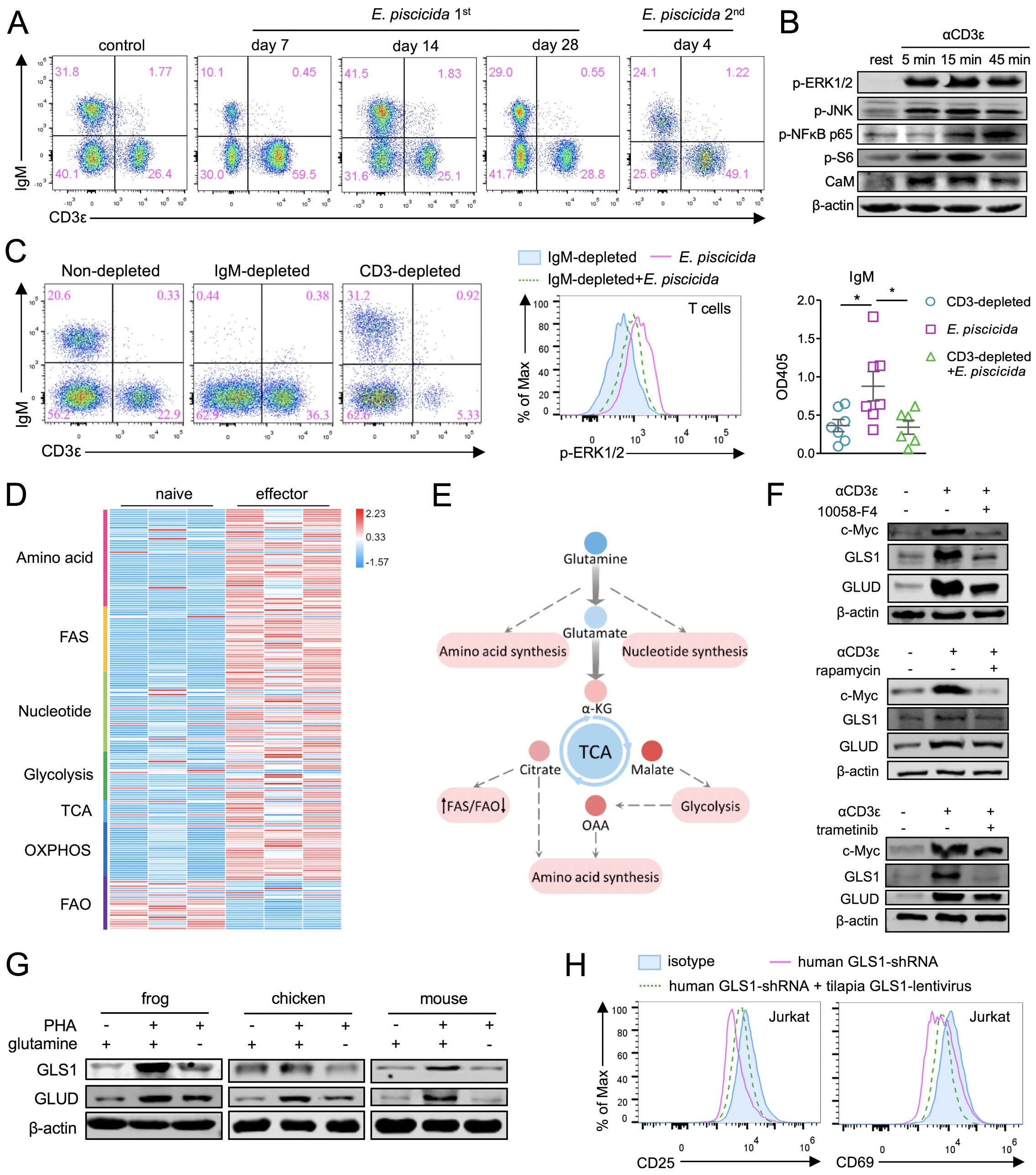
Glutamine metabolism regulates T cell immunity in teleosts and tetrapods
To analyze the regulatory mechanism of fish T-cell immunity, the authors conducted transcriptomic analysis. The results show that tilapia uses a transcriptional regulatory network similar to that of mammals to support its T-cell immunity in the evolutionary process. However, when the tilapia is affected by pathogen, the differentially expressed genes of T-cells are mainly concentrated in the metabolic pathways, and large-scale metabolic reorganization occurs.
As the reprogrammed metabolic programs can be directly or indirectly traced to glutamine catabolism, the research further studies the regulation of glutamine metabolism on tilapia T-cell immunity. The activated tilapia T-cells increases the utilization of glutamine; whereas the glutamine deprivation or inhibition of the glutaminase GLS1 results in impairment of T-cell proliferation and effector function. Further studies have confirmed that the mTORC1 and MAPK/ERK pathways have undertaken TCR signaling, regulated glutamine metabolism through c-Myc, and promoted T-cell immunity in tilapia.
Based on the similarities between tilapia and mammals in T-cell metabolism and reprogramming, the authors speculated that the regulation of T-cell immunity by glutamine metabolism may be a conserved strategy in the evolution of jawed vertebrates. The authors selected vertebrates at different evolutionary stages and analyzed the relationship between glutamine metabolism and T-cell immunity in order to verify this hypothesis.
The results show that tilapia, Xenopus laevis, chicken and mouse share a conserved c-Myc-driven glutamine metabolism to regulate T-cell immunity. Furthermore, the knockdown of c-Myc or GLS1 impaired the activation and proliferation of human Jurkat T-cells, whereas the reconstruction of the glutamine metabolic pathway using the corresponding molecules from tilapia can rescue the immunodeficiency of human T-cells. This discovery provides a new perspective for understanding the evolution of T-cell immunity, and also provides a potential way to intervene in human immunodeficiency.
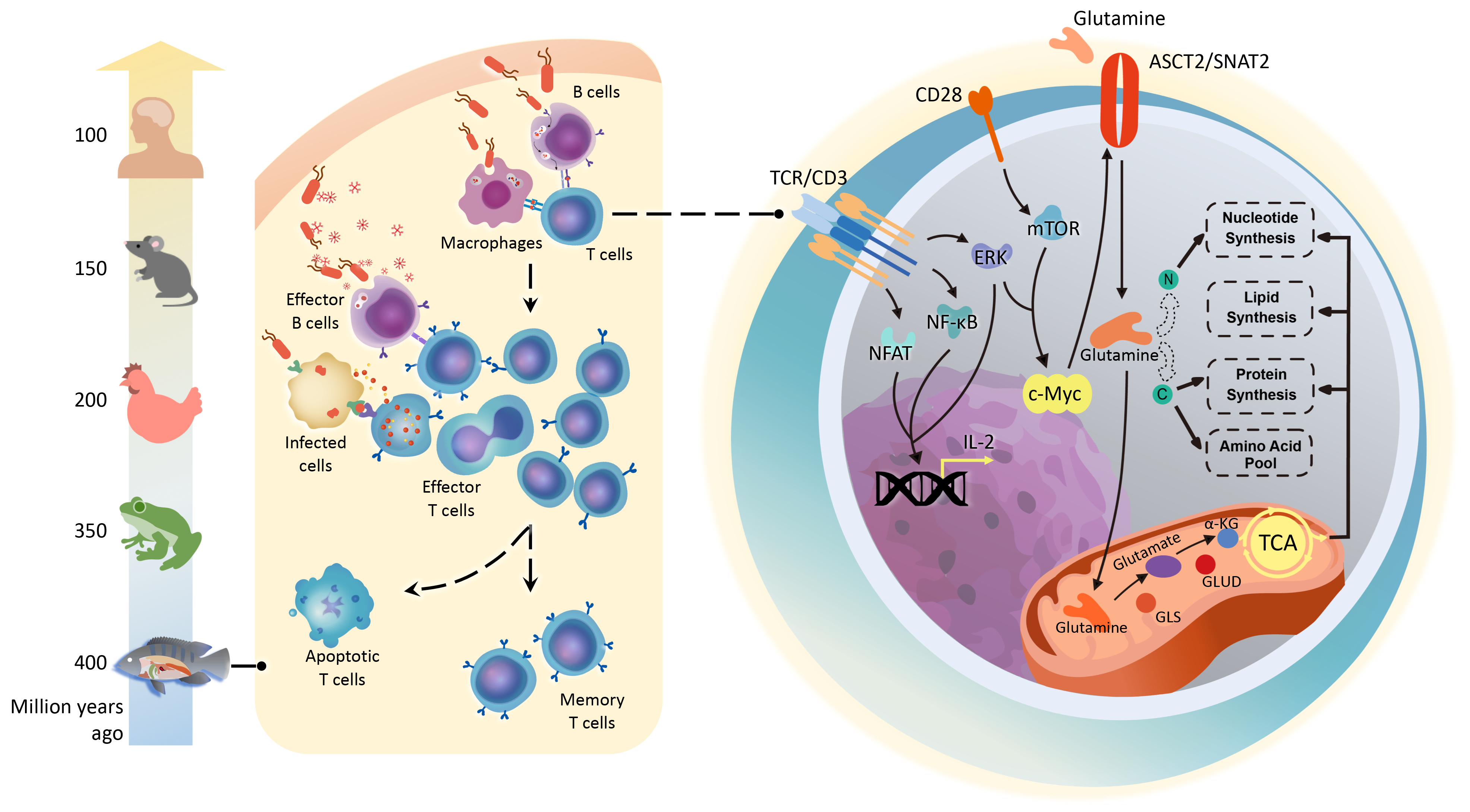
Glutamine metabolism is the molecular basis for similarities in T cell function between teleosts and tetrapods
In conclusion, this study has validated the hypothesis of stable evolution of T-cell immunity and proposes that conserved transcriptional regulatory networks and metabolic programs, especially glutamine catabolism, are the molecular basis responsible for the similar functions of T-cells between teleosts and tetrapods. The study was selected as the cover story [JM1] of Advanced Science.

Implication of the cover story picture: During the evolution (jumping the dragon) from the teleosts (carp) to the quadruped (dragon), T-cell immunity remains basically unchanged, and the conserved glutamine (molecular formula) metabolic pathway has laid the molecular foundation for this stable evolution. The picture was drawn by Jia Dianmo, a doctoral candidate from the College of Fine Arts, ECNU.

Doctoral candidate Li Kang, Associate Professor Wei Xiumei, postgraduate student Jiao Xinying from the School of Life Sciences of ECNU, and Dr. Deng Wenhai from Wenzhou Medical University are the co-first authors of this article, and Prof. Yang Jialong from ECNU is the corresponding author of this article. The research was funded by the National Natural Science Foundation of China Outstanding Youth Science Fund Project, and the General Program, etc.
Article linkage: https://doi.org/10.1002/advs.202201164
Copy editor: Melody Zhang, Mayfield Joshua Monroe
Editor: Yvaine Lyu